主な利点
-
レンチウイルスパッケージングサービス
配送時間は最短8~12営業日 -
正確な力価測定
導入後のqPCRにより、過大評価の懸念が排除されます。 -
プロフェッショナルな技術サポート
お問い合わせやご要望への回答は1営業日以内にお届けします。 -
強化された安全性
「自己不活性化」第3世代システムによりバイオセーフティが向上 -
ワンストップソリューション
ベクター設計とウイルスパッケージングから徹底的な分析とテストまで -
安全な配送
最適な効率を維持するためのドライアイスとコールドチェーン物流
サービスの詳細
| In vitro grade | In vivo grade | |
|---|---|---|
| Application | Cell culture | In vivo study |
| Quantity | 5E7 TU and up | 5E7 TU and up |
| Functional titer (post-transduction qPCR) |
≥1E+8TU/mL | ≥1E+8TU/mL |
| Physical titer (p24 or qPCR) |
≥ 2E+9 GC/mL ≥ 1E+10 LP/mL |
≥ 2E+9 GC/mL ≥ 1E+10 LP/mL |
| Purification | Crude | Sucrose gradient |
| QC (see add-on QC below) |
Post-transduction qPCR, fluorescence images if contain fluorescence | Post-transduction qPCR, fluorescence images if contain fluorescence |
| Timeline | Start from 7 business days | 3-4 weeks |
- レンチウイルスは 1E8 TU/mL 以上の濃度で提供されます。
- ウイルスパッケージングプロセスに有害な要素を持つ構築物については収量は保証されません。これには、毒性遺伝子、ウイルスパッケージングを阻害する遺伝子、二次構造を乱す RNA 完全性、配列再編成を起こしやすい配列、ヌクレオタンパク質、膜貫通タンパク質、受容体遺伝子、および LTR 間に 6.5kb を超える遺伝子セグメントなどが含まれますが、これらに限定されません。
品質管理
| Category | QC test | Description | Release standard |
|---|---|---|---|
| Titer | qPCR | qPCR quantifies the number of viral genome copies providing an accurate measure of the viral titer | Report titer |
| Post-transduction qPCR | Infection of cells followed by quantitative qPCR to determine the titer of lentiviruses | Report transduction unit | |
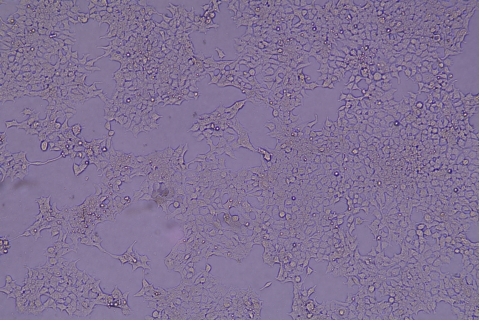
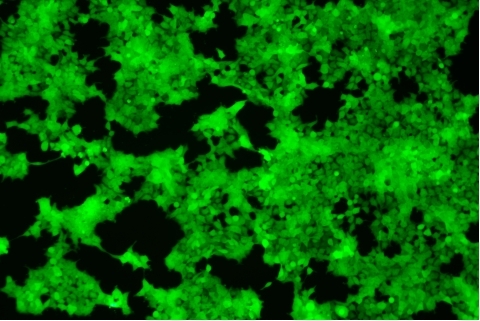
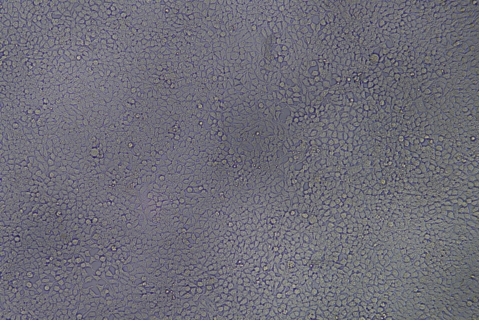
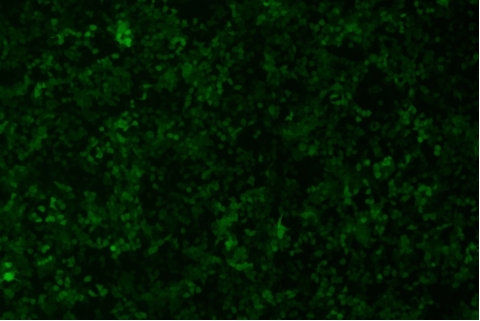
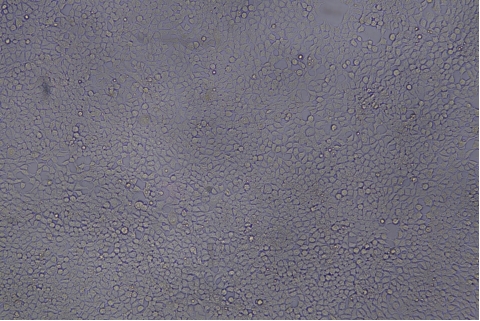
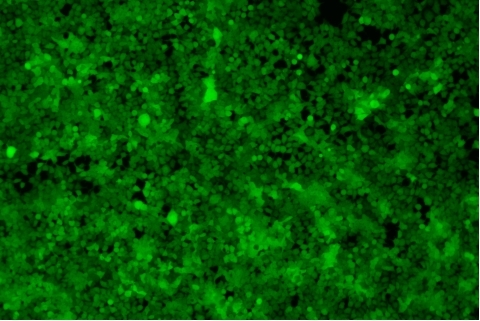
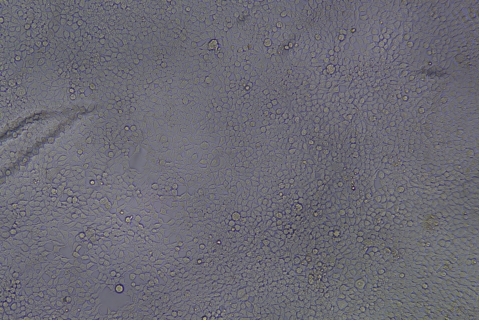
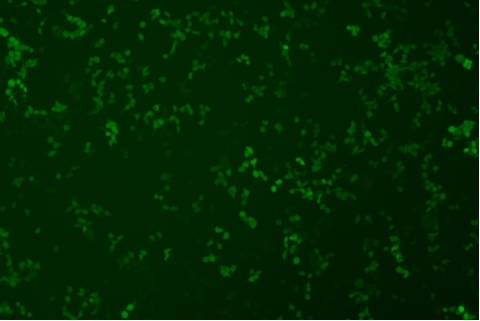
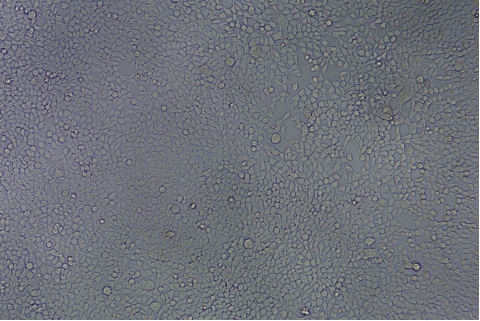
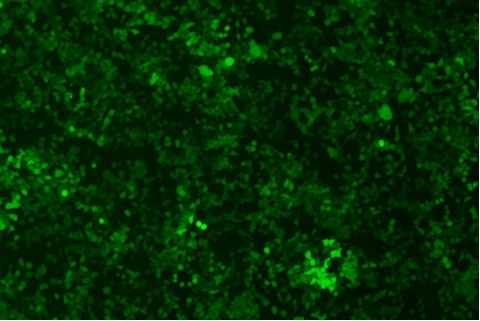
| Transduction test | Gradient dilution of infected cells and count of fluorescent cells | Provide bright field and fluorescent microscopy images | |
| p24 Elisa* | ELISA test for the p24 protein, a core capsid protein of HIV that is used to measure lentivirus titer | Report titer | |
| Contamination | Mycoplasma test* | Measure mycoplasma DNA by PCR | Guaranteed negative |
| Bioburden* | Plate count method: Quantification of live microorganisms (fungi, bacteria, etc.) using colony-forming units (CFUs) | No growth on the agarose plate | |
| Safety | Endotoxin LAL* | Measure endotoxin level by gel clot assay | Report |
* 追加料金の QC
レンチウイルスコントロールは必要ですか?
| Off-the-shelf lentivirus | Virus per vial | Titer |
|---|---|---|
| CAG.EGFP.T2A.Puro.WPRE | 2E7 TU | ≥1E+8TU/mL |
| CAG.EGFP.T2A.Bsd.WPRE | ||
| CAG.EGFP.T2A.Hygro.WPRE | ||
| CAG.mCherry.T2A.Puro.WPRE | ||
| CAG.mCherry.T2A.Bsd.WPRE | ||
| CAG.mCherry.T2A.Hygro.WPRE | ||
| EF1.SpCas9.T2A.Puro.WPRE | ||
| EF1.SpCas9.T2A.EGFP.WPRE | ||
| U6.NCshRNA.CAG.EGFP.T2A.Puro.WPRE | ||
| U6.NCshRNA.CAG.mCherry.T2A.Bsd.WPRE |
パフォーマンス
-
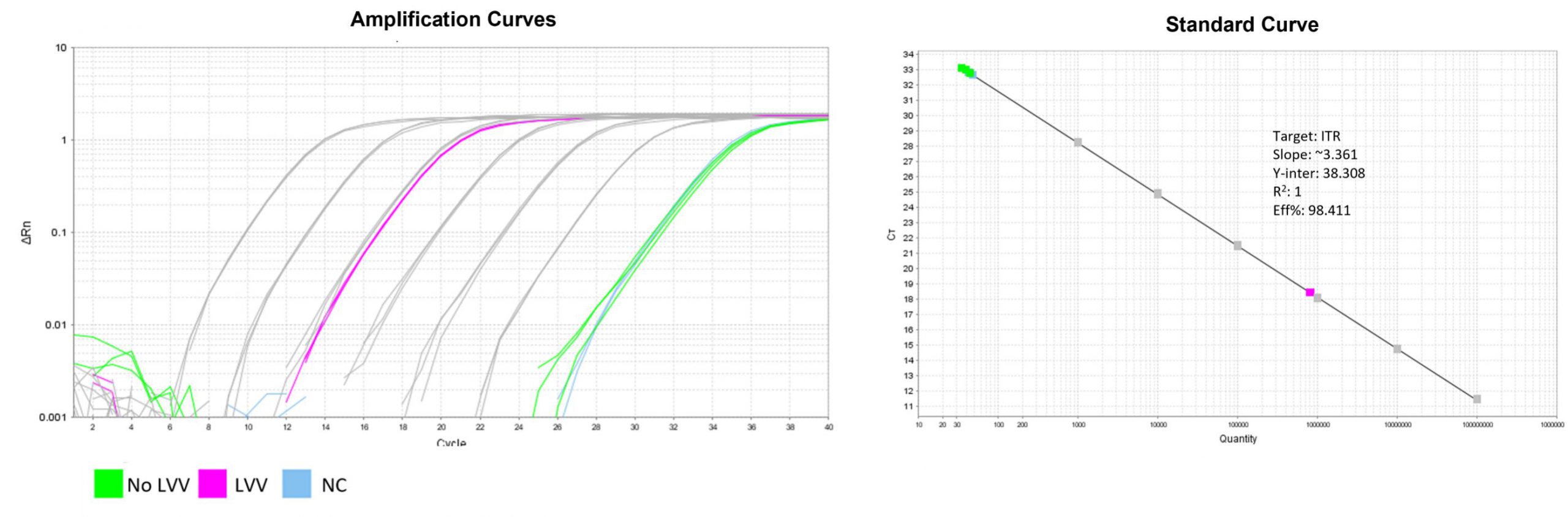
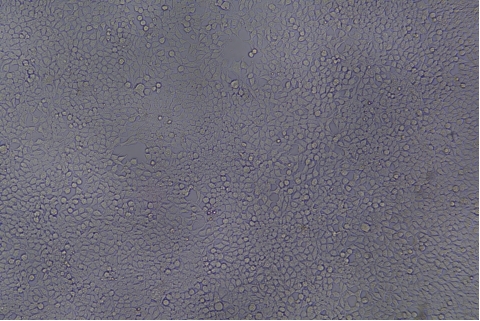
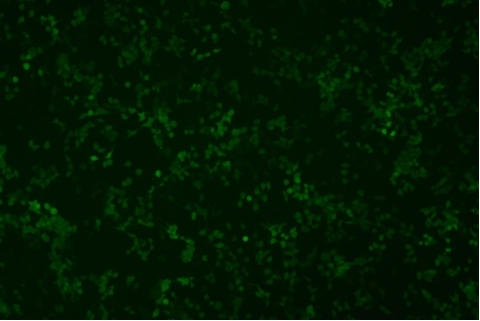
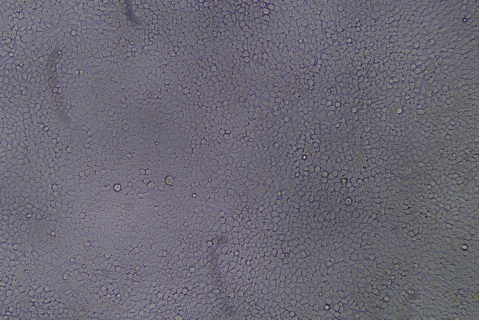
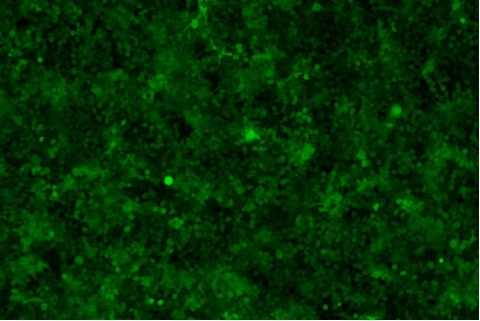
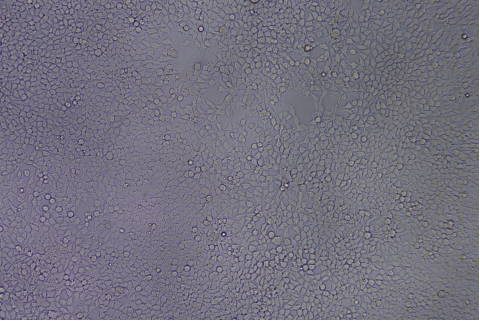
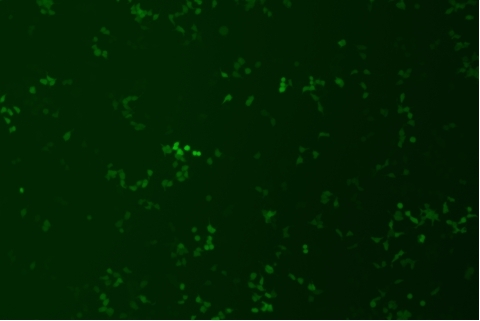
形質導入後力価 ロット間一貫性の保証
過大評価の心配はなくなりました
形質導入後 qPCR による正確な力価測定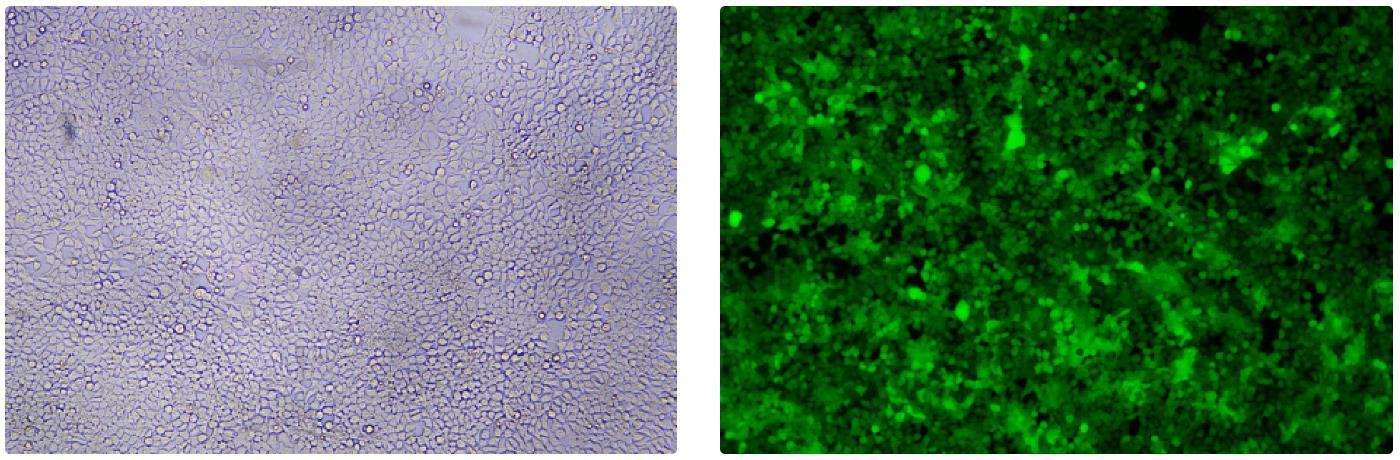 96 ウェルプレート、1.5E+04 cells/well、ウイルス 1.8μL、形質導入後 72 時間 QC テストデータの詳細
96 ウェルプレート、1.5E+04 cells/well、ウイルス 1.8μL、形質導入後 72 時間 QC テストデータの詳細 -
形質導入後力価の測定方法